Could Stem Cells Extend Human Lifespans?
From regenerating organs to curing incurable diseases, stem cells will be a big part of the future of medicine.
Welcome to Space Race, where we explore the impact of breakthroughs in science research on the 21st century. Subscribe for weekly breakdowns of the scientific breakthroughs that will shape the future.
In this week’s newsletter we discuss stem cells. While most cells in the human body have only one function, stem cells can transform into any type of cell. This gives them the huge potential to regenerate entire organs damaged by disease. We’ll discuss how stem cells work, how to get them/make them in the lab. Finally, we discuss their applications in an exciting new field of medicine called regenerative medicine that could cure diseases such as Alzheimer’s and Type 1 Diabetes.
One of the most intriguing biological puzzles is that of longevity — the challenge of increasing our lifespans. Modern medicine has already been incredibly successful at this, more than doubling average human life expectancy from a paltry 30 years before the 19th century to 70-80 years today. Given this success, it’s natural to wonder how we might achieve the next doubling of this number. One of the scientific breakthroughs which will play a huge part in the life extension story are stem cells.
What Are Stem Cells
The human body, being the exquisitely complex mechanism it is, has over 200 types of cells. Many of these cells are extremely specialised for one or at most a few tasks. For example, red blood cells have the protein haemoglobin and a disc-like shape which gives them a large surface area. Both of these optimise for oxygen absorption. Neurons on the other hand have a totally different structure. Instead of a round smooth disc, they have connectors called axons and dendrites. These allow it to optimise for electrical conduction and let signals travel at 430 km per hour (268 mph).
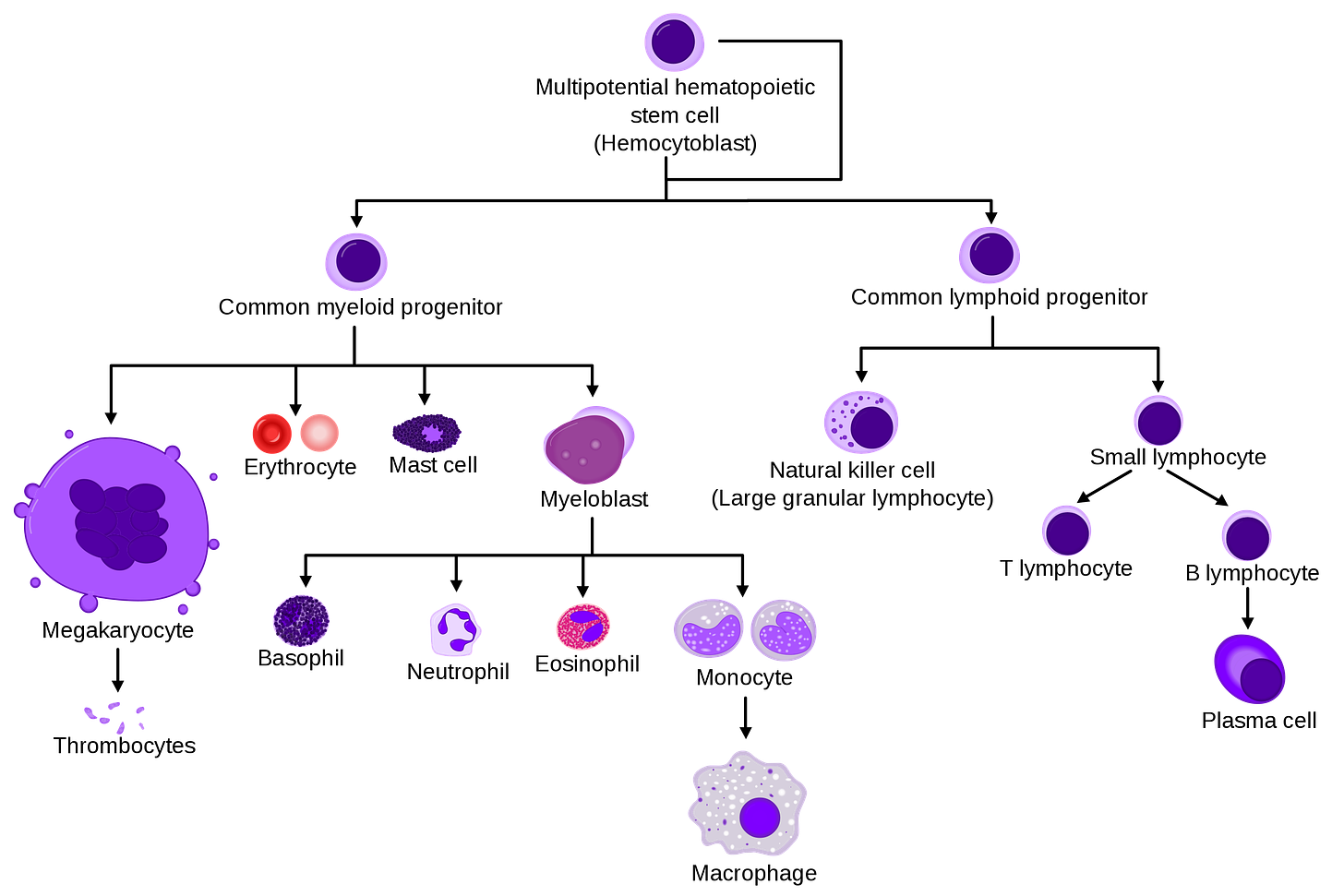
Stem cells are the opposite of specialised cells. They serve no specific function as they are but they can differentiate into various types of specialised cells. Because of this unique ability, they serve as an important regenerative/repair mechanism in the body. An important example of stem cells is the hematopoietic stem cell, which is produced by the bone marrow and differentiates into different types of blood cells such as red blood cells and white blood cells. The ability to specialise into different kind of cell types in a single family of cells is known as multipotency. Because of this ability, hematopoietic stem cell transplants have become a critical procedure for patients with cancers of the blood or bone marrow like leukemia.
Hematopoietic cells are one example of adult stem cells, which are stem cells that are found in fully grown humans. Another type are neural stem cells (NSCs), discovered in the 1960s, which were found to be able to differentiate into all of the specialised cells found in the nervous system. NSCs have so far been found in at least two parts of the adult brain: the subventricular zone and the hippocampus, which is involved in higher functions like learning, memory, and spatial coding. While these adult NSCs were a very important discovery, they are not present in sufficient quantities to repair serious damage to neurons in the broader brain. This, and the fact that neurons typically do not divide like other normal cells,1 is why neurological diseases like Alzheimer’s are so serious. Once it occurs, neuron damage is usually permanent.
While adults have some stem cells, for the most part their cells have fully specialised and any stem cells play a secondary role. Where stem cells play the main role is right at the start of human development, at the embryonic stage when all the organs and structures of the body need to be created. Since these initial cells need to specialise into all of the more than 200 different cell types, they are far more powerful in their ability to differentiate than adult stem cells. Instead of being only multipotent, embryonic stem cells (ESCs) are pluripotent, with the ability to differentiate into any type of human cell required in the embryo.2
Regenerative Medicine
Because they are more powerful than normal adult stem cells, pluripotent stem cells from embryos have incredible potential for medical applications. Understanding them has given rise to the field of regenerative medicine, which aims at restoring tissues and entire organs for patients suffering from severe injury or disease.
One example of a currently incurable disease that stem cells could impact is type 1 diabetes (T1D), which affects millions across the US. Normally, our pancreas makes a hormone called insulin to regulate our blood sugar level. The pancreatic cells that make insulin are called β cells. For reasons that are not well understood, in T1D the body’s immune system attacks and destroys its own β cells, resulting in no insulin production. The discovery of insulin by Frederick Banting in 1921 offered for the first time an ability to control this disease, but not a final cure for it. Banting himself cautioned at the end of his Nobel Prize lecture that “Insulin is not a cure for diabetes; it is a treatment.”
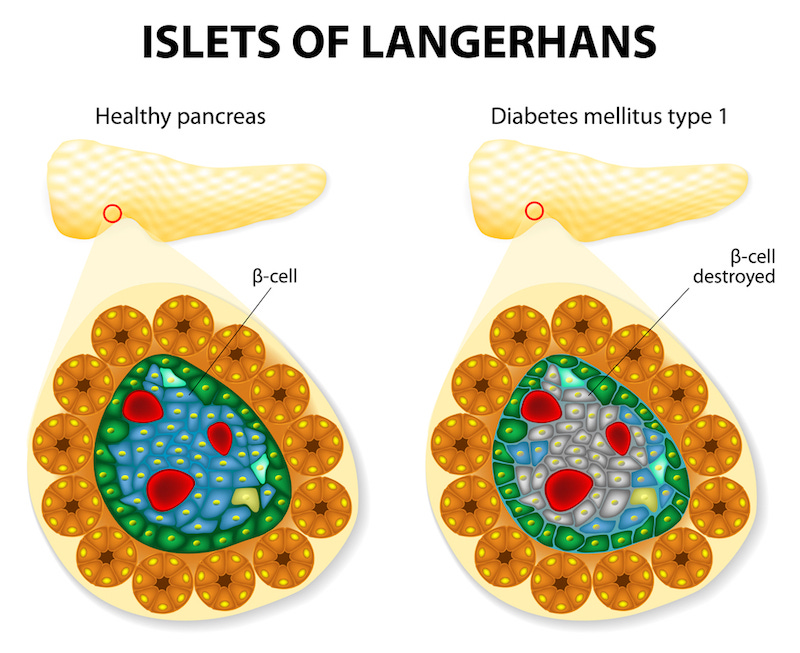
However, stem cells might offer a cure for T1D. Recent breakthroughs have demonstrated that β cells transplanted into T1D patients had the ability to successfully cure the disease. While this was an amazing discovery, one huge problem is the limited supply of donors. With pluripotent stem cells however, one could generate an effectively unlimited supply of β cells. Regenerative treatments for diabetes are still in their infancy but their therapeutic potential is already attracting huge amounts of interest.
One important question which must be answered before such transplant based approaches become widespread is immune rejection. While the immune system in T1D patients may not attack transplanted β cells just because they are β cells, it can still attack them because they are cells ultimately from a foreign body. This is the exact same process that occurs when a foreign virus or bacteria enters the body. Given that pluripotent ESCs cannot be produced by adult patients, any organs made from them will always be foreign and have the risk of immune rejection. This is a major obstacle to any potential application of ESC based regenerative healing.
In addition, ESC research also faces a huge non-scientific complication. Since harvesting stem cells from embryos often results in their destruction, research/treatments based on stem cells are plagued with difficult ethical issues.
Induced Pluripotent Stem Cells
But what if we could somehow “reprogram” mature specialised adult cells to become immature pluripotent stem cells? Then we could regenerate new tissues from the patient’s own cells and nullify the numerous ethical concerns around ESCs. In 2006, the Japanese scientist Shinya Yamanaka revolutionised the field by demonstrating that this was possible.
The key insight is to realise that even though each of our 200+ cell types is incredibly specialised for its unique function, they all contain exactly the same DNA. The specialisation rises from the fact that different types of cells are influenced to read different genes in our DNA by various proteins. One such class of proteins are transcription factors (TF). There are over 1600 types of transcription factors controlling what genes are expressed in our cells, which gives rise to the huge diversity of cell types in the body from one common set of genetic instructions in our body’s DNA.

Yamanaka and his colleagues showed that they could reprogram mature mouse cells by introducing 4 specific TFs called OCT4, SOX2, cMYC, and KLF4. When transplanted back into the mice, these reprogrammed cells were able to grow into all the different types of tissues as expected from pluripotent stem cells. In 2007, they were able to repeat this conversion with human cells using the same transcription factors. Stem cells created in this way are known as induced pluripotent stem cells or iPSCs.3
iPSCs obviously hold great promise. They offer each individual an unlimited supply of their own pluripotent stem cells to regenerate their own tissues. They also do not require any destruction or manipulation of embryos. All that’s needed is a sample of cells which can be easily extracted from the patient’s skin or blood. Free from immune rejection and ethical concerns, iPSCs would make regenerative transplants of all types possible.
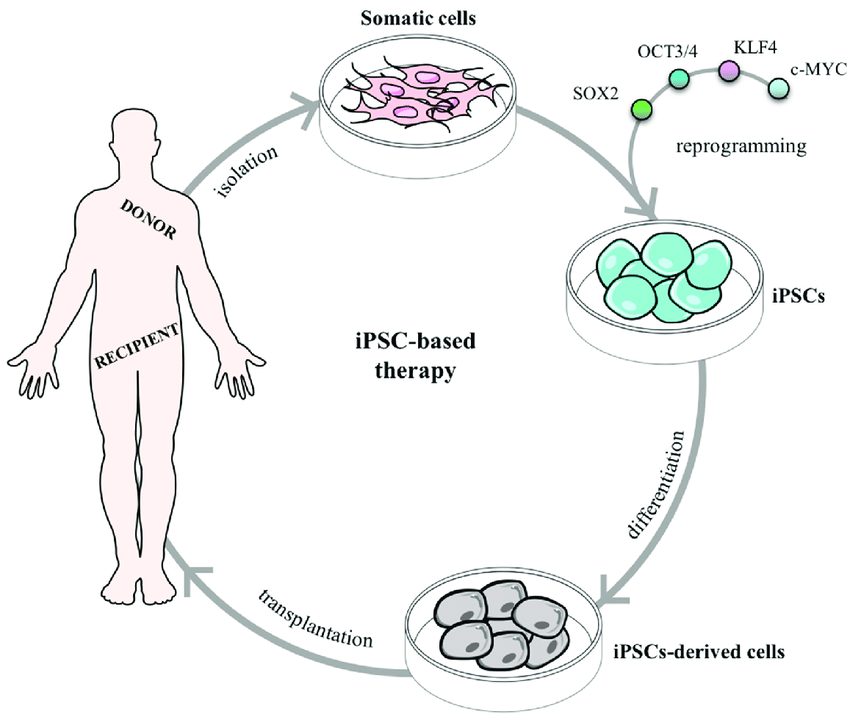
Although they solve the critical issues of ESCs, iPSCs present novel technical challenges that must be solved before they can be used with patients. One challenge is efficiency. Yamanaka’s method was successful at reprogramming only 0.01-0.1% of the normal cells used. This might be resolved fairly soon though as more recent approaches have demonstrated considerably higher efficiencies.
A more serious concern is tumorigenicity — the propensity of iPSCs to form cancerous tumours. ESCs are also tumorigenic to some degree, but iPSCs are predicted to have a greater ability to form tumours as a side effect of the reprogramming process. Quantifying and managing this tumorigenicity is still a very active area of research.
A promising application of iPSCs that doesn’t involve transplanting them directly into patients is using them as a platform for personalised drug discovery. To illustrate this, let’s return to the Alzheimer’s example. In Alzheimer’s disease, neurons degenerate at an accelerated pace for reasons that are not well understood. The inaccessibility of neurons in the brain makes it very difficult to perform research to discover the causes of the disease and test out therapeutic methods.
However, iPSCs generated from easily accessible skin or blood cells can be used to grow exact copies of a person’s neurons in a lab dish since they are derived from the same DNA. With this, scientists can investigate the patient’s unique genome and come up with therapies personalised to their condition, such as trying different drug combinations or identifying locations for precise CRISPR gene edits to reverse the neurological damage. Labs all over the world have already begun using iPSCs like this, such as the Early Drug Discovery Unit at McGill University.
Stem cells are clearly going to be a huge part of life extension 2.0. Their ability to differentiate into any type of cell gives us the incredible capacity of regenerating tissues and entire organs damaged by disease. Regenerative medicine has already shown glimpses of its power, such as its early success with Type 1 Diabetes. With the discovery of iPSCs, the huge challenge of immune rejection and ethical complications around embryonic harvesting are also removed, although some serious challenges like tumorigenicity remain and are actively being researched. Finally, the combination of advances in stem cells and gene sequencing/editing enables new methods of personalised drug discovery which could help us cure diseases like Alzheimers. Overcoming such “incurable” diseases will undoubtedly be a critical step towards the next doubling of human lifespan.
Because of the incredibly complex nature of the network of neurons, adding an extra neuron would disturb the precise electrical pathways already connected.
Technically, pluripotent cells can specialise into any type of cell except those of the placenta. Cells which can specialise into every single type of cell, such as the initial fertilised egg, are called totipotent.
Yamanaka apparently named iPSCs with a lower case “i” due to the popularity of Apple’s iPod.