CRISPR Part II: Evolution or Extinction
What happens when humans break free of natural selection?
Welcome to Space Race, where we explore the impact of breakthroughs in science research on the intensifying global contest of superpowers in the 21st century. Subscribe for weekly breakdowns of the scientific breakthroughs that will shape the future.
Today’s newsletter is part 2 of our series on CRISPR, an amazing technology that can edit genes with unmatched precision and speed. If you missed it, you can read part 1 here. This week’s article focuses on human applications of CRISPR. We’ll see how CRISPR gene editing could cure previously incurable diseases such as HIV-AIDS. If used to make inheritable changes, CRISPR could also allow us to control our own evolutionary destiny. We’ll explore the possible consequences having such control over natural selection itself, including the deep ethical dilemmas such immense power raises.
By now, we have a basic understanding of how CRISPR works, and have seen some examples of its incredible impact in domains such as agriculture. However, the stakes of CRISPR gene editing are highest when the intended recipients are not staple crops or malaria-carrying mosquitoes but humans.
CRISPR’s human applications can be broken down into two categories. The first are somatic applications. These target somatic cells, which includes all cell types except the egg and sperm. These cells can be targeted to cure genetic diseases, e.g. red blood cells can be edited with CRISPR as a therapy for sickle cell disease. As we will see, somatic applications have the potential to achieve incredible cures for diseases previously thought incurable.
The second class of CRISPR applications are germline applications. Germline cells are the egg and sperm, which are extremely special as they are the only cells which pass on genetic material to the next generation. It is here that CRISPR truly grants us the power to control our own evolutionary destiny. However, the ability to dictate the genetics of tomorrow’s humans is not a privilege to be taken lightly. Even small changes to the germline might amplify into species wide unforeseen consequences. Beyond catastrophic mistakes, one cannot forget the dangerous allure of using CRISPR as a eugenic tool. Because of such concerns, germline editing is littered with profound ethical dilemmas that we must think about before using CRISPR to make inheritable changes.
Somatic Applications
Somatic gene therapy with CRISPR targets body cells which are affected by genetic diseases such as sickle cell disease or cystic fibrosis. If the DNA segment that gives rise to the abnormal behaviour is known, it can be cut using CRISPR and replaced with the correct gene if necessary. Of course, if a person has a genetic disease, every single cell in the body will cary the defective DNA but it is clearly impossible to CRISPR edit all of the trillions of cells in the human body. However, that much hard work isn’t necessary. Since the the defective genes’ effects only manifest in particular cells, it is sufficient to simply edit the affected cells to remove the effects of the genetic disease.
Sickle cell disease is a good case study of a somatic application of CRISPR that is already poised to have a broader impact. This disease is caused when a person inherits two abnormal copies of the β-globin gene — one from each parent — which exists in chromosome 11 of the human genome. This gene causes sickle cell disease, due to which red blood cells become deformed into a sickle shape instead of their usual disc shape.1 Symptoms include sharp pain due to the misshapen cells trying to squeeze through veins and anaemia since the deformed blood cells cannot transport oxygen as well around the body. Millions around the world have sickle cell disease, and many more are inadvertent carriers of one abnormal copy of the β-globin gene, putting their children at risk of it.

While sickle cell’s symptoms can be managed with medication and other methods, the only option available to actually cure the disease up to now was costly bone marrow transplants. Even if a patient is able to afford this procedure, it is extremely risky as the body might suspect that the transplanted cells are invaders and mount an immune response against them. It is also very possible that no donor is found, in which case transplants are out of the question.
However, since the exact gene that gives rise to the sickle cell mutation is known, CRISPR can potentially supply a permanent cure. Instead of relying on an external donor for healthy bone marrow cells, one can instead take bone marrow from the patient themselves, edit the defective β-globin gene and reinsert it. This way, there is no risk of not finding a donor, and of the body rejecting the donated cells because the patient can simply be their own donor. Once reinserted, the repaired bone marrow cells can produce the correct type of red blood cells, effectively curing the disease.
Another potential application of CRISPR is as a permanent cure for HIV-AIDS. While HIV affects populations around the globe, a very small number of people have a natural immunity to it. The cause of this immunity is the deletion of 32 DNA bases in the gene that codes for the protein CCR5. The CCR5 protein is usually located on the surface of white blood cells and is what HIV attaches to when infecting an individual. In individuals that naturally don’t produce CCR5, HIV has no way to begin its infection since it has no protein to latch onto. The lack of this protein isn’t yet known to cause any other adverse effects beyond a greater susceptibility to West Nile virus. Given this, the basic contours of a CRISPR based cure appear naturally. One theoretically just needs to make a precision cut in the CCR5 gene to deactivate it. This would provide natural immunity to HIV.
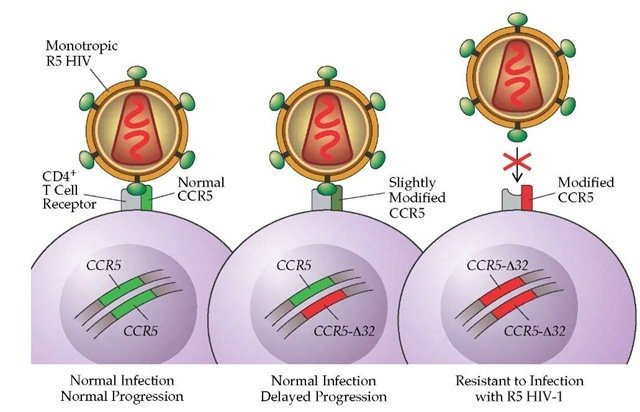
It is clear that somatic editing has huge potential, but as with any new technology and its applications, we must be sure that its use doesn’t raise new complications. For example, in the HIV cures, scientists must be completely confident that deactivating the CCR5 gene really does not have any other adverse effects. After all, there is a reason that humans evolved that gene in the first place.
It is likely to be a long time since such CRISPR based cures are widely available since they must go through rigorous clinical trials. However progress has been rapid and CRISPR based therapies have already demonstrated success against sickle cell. Recently, such therapies that target both sickle cell disease and HIV have gained approval for further trials from the US Food and Drug Administration. If successful in these, there is little doubt that CRISPR based therapy will become an indispensable part of doctors’ toolkits to cure previously incurable diseases.
Germline Editing
Any new technology carries risk reward tradeoffs. Somatic applications of CRISPR have the potential to cure incurable diseases, but could also cause unknown complications. The human genome is an incredibly complicated system and it’s not difficult to imagine that well intentioned actors might simply overlook some small but vital function of a gene before editing it. The stakes are high, but a natural failsafe is that any negative effects are strictly local — they are only restricted to the individual patient whose genes are edited.
In germline editing, where we edit the egg and sperm cell DNA, this failsafe disappears and the stakes elevate exponentially. If there is any mutation effected by CRISPR — good or bad — offspring could inherit it. Now the impact is not localised and could spread throughout the human population. Removing this final check on our power to edit the genome is what gives us the ability to control evolution. With this greater possible reward comes an equally greater risk.
The most apparent danger is a well intentioned mutation having unforeseen harmful effects. Suppose one edits CCR5 in the germline so that children inherit HIV immunity, but it is later discovered that the CCR5 mutation might contribute to lower life expectancy. This raises immensely complicated questions. Is there a point where the tradeoff in life expectancy is worth the immunity to HIV? Who should have a say in such decisions? One answer might be that of course the prospective parents should decide. However, it is hardly likely that anyone except those familiar with the cutting edge of research will have the required knowledge to debate this tradeoff in an informed manner. What if the gene edit is harmful in the long run — who takes responsibility if/when it spreads throughout society? These are all difficult questions with few easy answers. The fact that CRISPR is still a very active subject of research with possibly many unknown nuances complicates this all further.
Scientists are often wary of giving such ethical dilemmas the proper debate that they deserve. To some degree, this is understandable. It is often difficult to devote time to think about broader impacts of a technology when it is a struggle to simply develop it in the first place. When there is no guarantee that what you are working on will even be technically sound, it can be counterproductive wishful thinking to entertain grandiose ideas of broader applications.
However, by now CRISPR has demonstrated that it has very real applications with huge impacts. As such, it is imperative that we pause and think about the ethical questions that arise when this immensely disruptive technology interfaces with society. It was exactly due to this that Jennifer Doudna herself called for a moratorium on germline editing with CRISPR until a more thorough dialogue had taken place on the ethical conflicts it raises.
To see just how real the problems posed by CRISPR are, consider the case of Chinese researcher He Jiankui. In 2018, He used CRISPR to edit the CCR5 gene in human embryos to try confer genetic resistance to HIV. This work, which was conducted in secrecy, created tremendous controversy when it was made public by the MIT Technology Review. Although He claimed that the children, known as Lulu and Nana, were born healthy, the global outrage and concerns for the girls’ health prompted China to hand him a $430,000 fine and three year prison sentence. The fact that recent research has also suggested that the CCR5 edit may be correlated to lower life expectancy no doubt complicates this whole episode further. This is more or less an exact replica of the toy example we considered above, and shows that these aren’t just abstract debates but real questions about real problems which already exist.

CRISPR & Inequality
Let’s put on our rose tinted glasses and suppose that we do manage to make germline edits which are guaranteed to not have unexpected effects. Surely such a case is the dream scenario? We could give our children whatever traits we desired — high intelligence, super strength, taller height, or perfect pitch. Putting aside the very obvious eugenic concerns here, there is another question that emerges when we realise that CRISPR, like any new technology, must interact with human society. That is the issue of equality of access.
Like most new technology, it is likely that CRISPR will be disproportionately used by the wealthy — at least initially. However, if the initial users can augment their genetic lines with any advantageous traits they want (assuming the dream scenario of no unexpected consequences) then we risk transforming the economic inequalities that already exist into permanent genetic ones.
Inequality stems largely from the fact that while talent is equally distributed around the world, opportunity is not. Those who have the chance to capitalise on their gifts generally tend to do so and make something of themselves. If CRISPR skews the distribution of talent as well, this is perhaps the most extreme and dystopian iteration of the rich getting richer. It would be as if our societal class was imprinted into our genetics to create a de facto caste system. Nothing could be more abhorrent and antithetical to the notion of a fair society.
Of course, just because germline editing can have negative, possibly dystopian, effects doesn’t mean it should be outright banned. There are undoubtedly huge benefits to be realised. For example, germline editing could enable parents with inheritable genetic diseases to have completely healthy babies. From an idealistic perspective, it can be argued that if a CRISPR edit has the chance to change someone’s life, then maybe it would be ethically immoral to not even consider trying it. From a realistic viewpoint, if we impose outright bans on something as revolutionary as germline editing, rogue elements will pursue that research anyway under the cover of secrecy, possibly leading to more dangerous outcomes. The He Jiankui affair is a clear example of that. To avoid this, germline research should be supported, but within a robust regulatory regime whose rules must be decided only after a thorough discussion with all stakeholders.
By now, it is clear that to say CRISPR is revolutionary is not indulging in gratuitous hyperbole. This technology offers us no less than the ability to edit the code of life itself. With such huge power comes immense responsibility and nowhere is this more true than when we inevitably start CRISPR editing our own genomes. The obvious opportunity for CRISPR in humans is to cure currently incurable diseases, such as sickle cell and HIV-AIDS. The main risk here is to make sure that the edits made do not have any inadvertent consequences, but this risk will likely drop given the speed with which genomics is moving today.
A second, far grander opportunity is to take control of inheritance through germline editing and dictate our own evolutionary trajectory. One might envision a future in which kids have been CRISPRed to remove genetic diseases from their genomes, or have genes inserted for greater intelligence. Of course, breaking free of the natural selection’s guiding hand like this poses tremendous risks of its own, such as corrupting the human genome through a bad edit or solidifying society’s class inequalities into permanent genetic ones. These risk-reward tradeoffs in turn present huge ethical dilemmas. Despite these significant obstacles, putting our heads into the sand and banning CRISPR outright is not an option. This technology is too powerful and the potential for improving lives is simply too high. The only sensible path forward is to encourage CRISPR research, but in a way such that scientists can both be innovative but also remain accountable to the public. Striking this golden balance won’t be easy, but it is absolutely vital if we are to avoid the mistakes of past revolutionary technologies.
This occurs because the defective β-globin genes interfere with the normal production of haemoglobin, a very important chemical that allows red blood cells to bind to oxygen. The mutated shape of haemoglobin molecules gives rise to the sickle cell shape of the red blood cells.