Everything You Wanted To Know About Superconductors
Including some thoughts on the (supposed) room temperature superconductor

Recently, the internet has been taken by storm with the news of a possibly revolutionary scientific discovery: the discovery of a room temperature superconductor. The news has created a frenzy as Twitter pundits breathlessly speculate that it’s only a matter of time before we have ultra-efficient power transmission lines, incredible new efficient batteries, and even desktop quantum computers. Just as we’re all still coming to terms with semiconductors as a result of the US-China chip war, it might be that superconductors open a new front on the technological frontier. As someone who builds superconducting electromagnetic resonators for a living, I’m thrilled to see superconductivity exciting so many people. So what exactly is a superconductor, what’s so special about room temperature, and is the Korean discovery real?
Superconductivity Explained
As I explained in my post on semiconductors, one of the fundamental classifications of materials is whether they conduct electricity or not. While metals like copper conduct electricity, insulators like plastic do not. What makes metals good conductors is the fact that their electrons are not tightly bound to their atoms but are free to move around. These are called delocalised electrons and their motion is what allows electrical current to flow. Insulators do not have delocalised electrons, so no current can flow.
However, even conductors have some intrinsic opposition to the flow of electric current. To see why this is, imagine you have a wire of copper. For the electrons to flow from one end to the other, they have to go through the lattice of copper atoms in the wire. It is extremely unlikely that electrons will travel in a perfect unimpeded straight line through the copper, and far more likely that they will bounce around and collide with copper atoms on their way. These collisions cause the electrons to give up some energy which slows them and so reduces the flow of electric current. This energy given up by electrons is the source of the heating that happens when wires carry electric current. For example, electrical transmission lines operate at temperatures of around 100 degrees celsius, which is hot enough to boil water. Impressive, but ultimately this is wasted energy since this heat can’t be used to do useful work.
The tendency of a material to oppose electric current is called its electrical resistance. While insulators have basically infinite resistance, conductors like copper have a small but nonzero value. For a given material, the resistance also usually depends on the temperature.
In 1911, the Dutch physicist Heike Kamerlingh Onnes was doing an experiment to measure how temperature affected the resistance of mercury. He observed a gradual decline as he cooled the sample, but when it was cooled to 4.2K (-269°C, -452°F)1 its resistance immediately and suddenly dropped straight to zero. Onnes had discovered the first superconductor.

The behaviour that Onnes observed is an example of what physicists call a phase transition. Water turning into ice or steam are also examples of phase transitions. Just like water turns into ice at exactly 0°C (32°F) and into steam at exactly 100°C (212°F), mercury goes from its normal to its superconducting phase at exactly -269°C. The temperature at which a metal suddenly loses all resistance and becomes a superconductor is known as the critical temperature (known in the lingo as Tc).
Following Onnes’ discovery, many more metals were found to be superconducting. Examples of everyday metals that are superconductors include tin (Tc = 3.7K), aluminum (Tc = 1.2K), and lead (Tc = 7.2K). One of my favourite superconductors is niobium (Tc = 9.2K), which I use all the time along with aluminum in my own research.

What explains this seemingly magical transition to a superconducting state? It’s very complicated but in a nutshell, this is a manifestation of the bizarre rules of quantum mechanics. To oversimplify a lot, at the critical temperature, the electrons pair up with each other forming what are called Cooper pairs. These Cooper pairs act as one effective particle instead of two separate electrons. A crucial difference between Cooper pairs and individual electrons is that Cooper pairs can all become highly “mixed” with each other and form a collective synchronised sea of particles called a Bose-Einstein Condensate. The key idea is that since all the pairs behave collectively now, to break a single Cooper pair we have to break all Cooper pairs. So the energy required to break one pair of electrons and deflect them off their path is the same as that needed to break all pairs of electrons. By acting together, the electron pairs resist any “kick” from the atoms in the metal or any other source and travel unimpeded. Therefore current will flow freely and the material will have zero electrical resistance.2
Applications of Superconductivity
So what are the potential applications of this? Considering that electrical resistance is a huge source of wasted energy in almost any application where electricity is used, superconductors could theoretically have applications throughout society. Examples include the aforementioned electrical transmission lines and batteries. One awesome application that dovetails with one of the most consequential contemporary technologies would be using superconductors to make energy efficient AI chips. That would make it easier to scale already vast AI models like GPT-4.
Unfortunately in practice, because of the low temperatures needed to achieve superconductivity, they remain far less widespread than their potential might imply. Today, if you want to use superconductors, you need a way to get them superconducting which involves a very heavy accompanying investment in cryogenics.
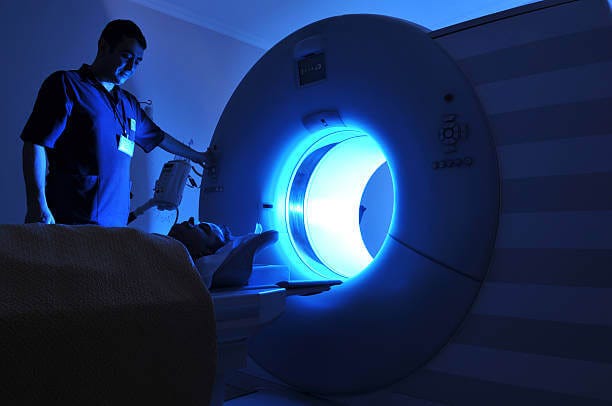
The current biggest market for superconductors is in Magnetic Resonance Imaging (MRI). MRI is the most sensitive imaging technology available for the human body, so no surprise it’s a huge $8B market. To achieve high imaging resolution, MRI machines require very large magnetic fields up to 100,000x stronger than the Earth’s field. Magnetic fields are generated by electric currents, so to make such large fields we need huge currents of around 100 amperes. For perspective, 0.01A is more than enough to kill a human. If you tried to run that current through an MRI magnet made of copper wire, the heat produced would destroy the machine, and probably the hospital also.3 Instead, MRI magnets use superconducting wire made of niobium-titanium alloy (Tc = 10K). To keep the wires cold, MRI systems use liquefied helium (T = 4.2K), which is expensive at $30-50 per litre but still more economical than a blown up hospital.
Another potential use is in Maglev trains. These trains use powerful magnets to levitate trains above tracks. Without the nuisance of friction, the train can travel much faster.
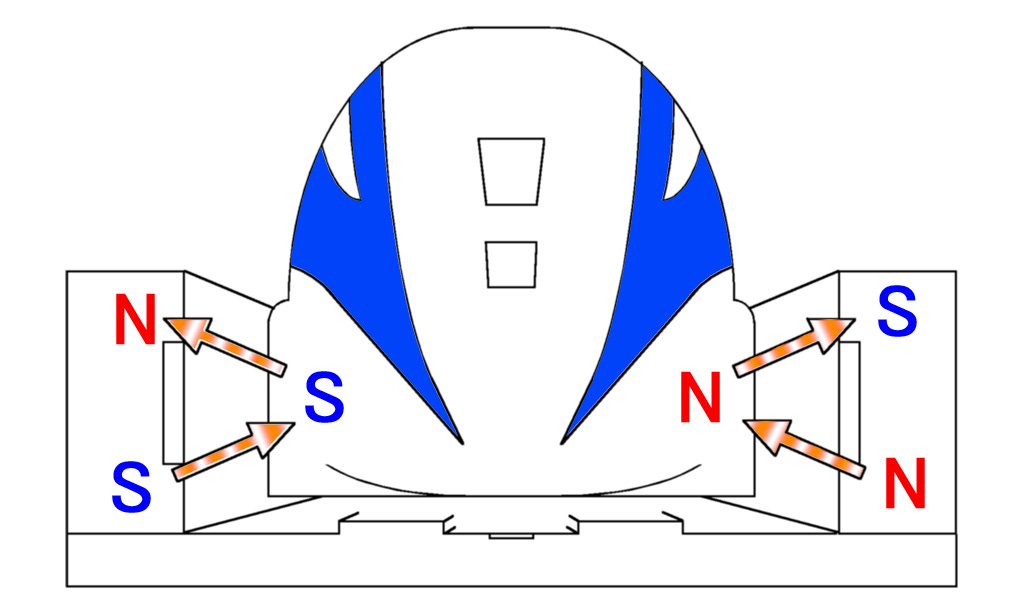
As with MRI machines, using normal resistive wiring would generate a lot of heat so superconductors provide a solution. The Central Japan Railway Company has demonstrated a prototype seven car train on a track using superconducting magnets in 2015 that reached a record 603 km/h (375 mph). Their SCMaglev system is currently approved for use between Tokyo and Nagoya by 2027 and to Osaka by 2037.
Another use of superconductors closer to the technological frontier is in quantum computing. Using superconductors is one way to make quantum bits (qubits). Quantum chips frequently use aluminum and niobium traces to make connections between electrical components. The use of aluminum with Tc = 1.2K means that instead of liquid helium, superconducting quantum computers require the most state of the art cooling systems: dilution refrigerators. At the press of a button, modern dilution fridges cool down to 0.01K, hundreds of times colder than outer space. All for the price of a cool couple of million dollars.

The Holy Grail: High Temperature Superconductivity
So superconductors have the potential to be incredibly useful, but the required investment in cryogenics makes most applications uneconomical. If we really want to realise incredible applications like ultra-efficient power transmission lines and AI chips, we need to decouple superconductors from cryogenics. This is why one of the holy grails of modern physics is the creation of a room temperature superconductor.
There is no theoretical reason why one can’t exist, and there’s already been a lot of progress on this front. Many high temperature superconductors are exotic ceramic materials made of a mixture of many elements, like Yttrium-Barium-Copper-Oxide (YBCO) which has a critical temperature around 93K (-180.15°C). That’s still far from room temperature, but it’s high enough that we can reach it using liquid nitrogen (77K), which is much cheaper than liquid helium at $0.51-1.78 per litre.
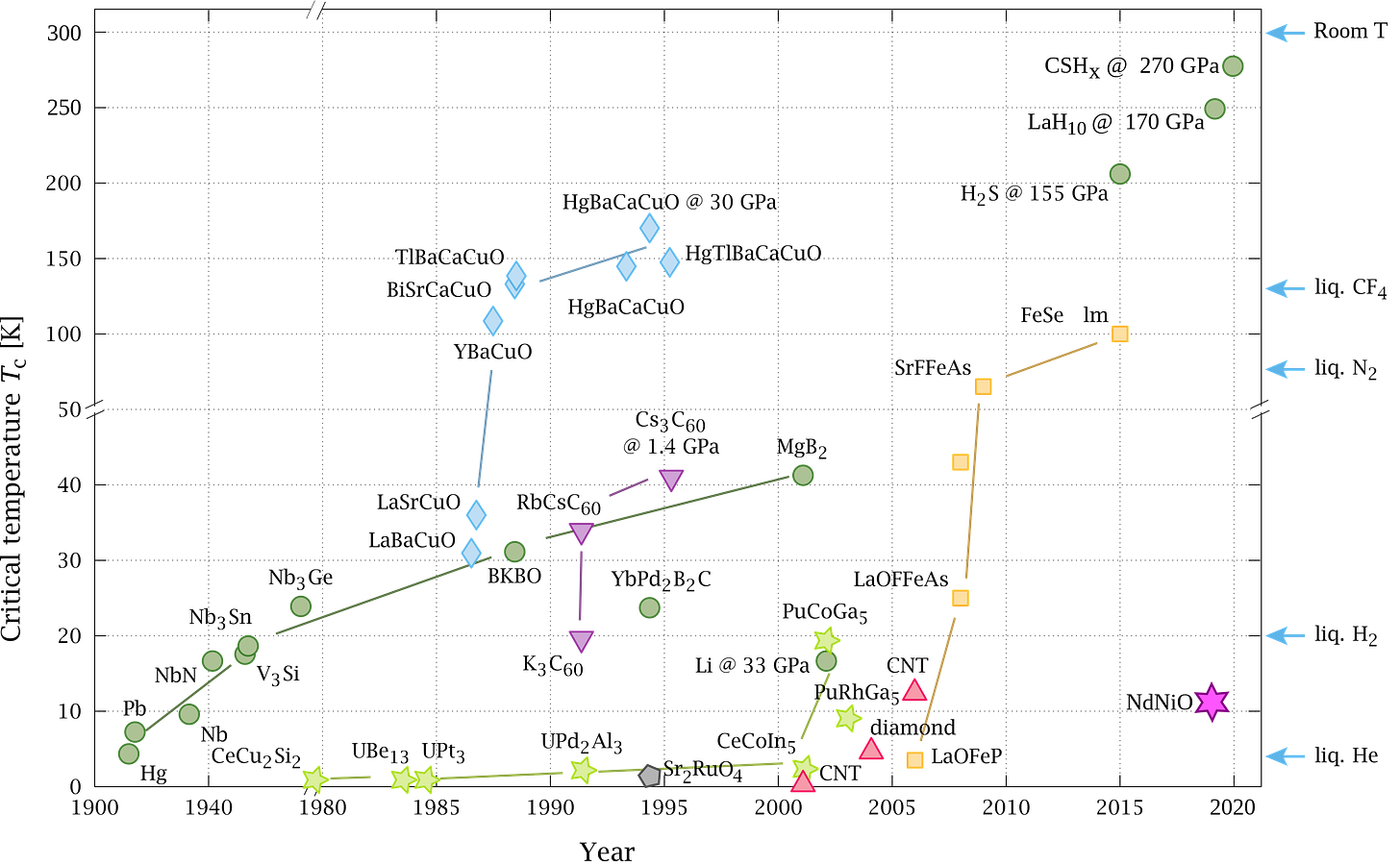
However, many such ceramic superconductors like YBCO are brittle and difficult to work with. Contrast this with a superconductor like aluminum, which even a grad student can machine structures out of or deposit on a chip without too much difficulty. Given this, up till now the lower cost of higher critical temperatures hasn’t been enough to offset the higher costs of producing the exotic high temperature superconductors in the first place, so they haven’t really spread.
LK-99 and The Korea Experiment
Since a room temperature superconductor is so high stakes — with a guaranteed immediate Nobel prize — there are very frequent claims of having realised it in the lab. Up till now all such claims have always been unable to stand up to the test of independent replication. For instance just earlier this year, the physics community was rocked by a claim of “near ambient superconductivity” by Ranga Dias in the prestigious journal Nature. He claimed he had made a superconductor with Tc=294K (21°C, 70°F) and a pressure of roughly 10,000 atmospheres. Dias has a history of making sensational superconductivity claims but researchers have never been able to replicate his results. He has also had multiple papers retracted for faking his data.
Unfortunately, since the prize is so great, the quest for a room temperature superconductor leads to papers with data that fails to replicate. It’s always because either the authors rushed their experiments and made some mistakes or deliberately manipulated their results. It’s in this context we have to judge the recent claim from Korea. The Korean team claims to have made a room temperature superconductor at normal atmospheric pressure, so it’s an even more sensational discovery than Dias’ fraudulent one would have been. So is it legitimate?
Let me preface my comments by saying that while I use superconductors extensively in my own research, I’m not an expert in the materials science behind them. As usual, the true test will be independent replication. The recipe for their LK-99 material seems easy enough to recreate. That being said, I’m not optimistic. Off the bat, the Korea team doesn’t report an actual Tc for their material, claiming only that it’s larger than 400K. This is really unusual since a superconducting transition is pretty sharp and well defined at a fixed characteristic temperature.
Beyond zero resistance, there is another observation necessary to confirm superconductivity: the Meissner effect. Basically as a result of all the quantum weirdness, superconductors also block out magnetic fields. This means that if you put a magnet above a superconductor, it will necessarily levitate. The Korea team put out a video of this, but I’m not so sure it’s convincing because the levitating material is never completely clear off the surface of the superconducting sample. Compare that video to the one below, where the levitation is much cleaner.
These are pretty qualitative and vague objections, but since I’m not an expert I hesitate to make any more precise comments on their data. On the other hand, there have been some encouraging theoretical calculations showing that LK-99’s electron energy level structure could support superconductivity. Nevertheless most superconductivity experts are urging caution. We’ve seen too many of these claims in the past to be naive about this one. As usual, we must wait for experimental replication to conclusively settle the debate.
However just because it hasn’t worked yet doesn’t mean we should stop trying. There is no good physics reason why superconductors can’t exist at high temperatures. As long as researchers don’t engage in deliberate malfeasance, every single attempt at making a room temperature superconductor is worth it simply because of the incredible outsized contribution it would have on society. Breaking superconductors free of their cryogenic cage would catalyse basically every technology we use today. It would truly be revolutionary.
4.2K is roughly -269 degrees celsius. 0K (-273.15°C), also known as Absolute Zero, is the coldest possible temperature according to the laws of thermodynamics.
This is known as BCS Theory after the physicists who came up with it (Bardeen, Cooper and Schrieffer). BCS explains most of the superconductors we know, but there are some which cannot be explained by BCS and require other theories.
For fun, let’s guesstimate this. Let’s say an MRI magnet is a coil of wire with diameter and length roughly 1m. The magnetic field strength is proportional to the number of turns on the coil, and an MRI magnet has around 10,000 turns. Assuming the wire turns are all stacked end to end, the wire cross section diameter is 1/10,000 = 0.0001m. Finally, copper has a resistivity of ρ = 1.68 x 10^-8 Ωm. The resistance for this wire ends up being ~70 kΩ. The formula for heating power is P = I^2 R. With a current of I = 100 A and R = 70 kΩ, we get P = 0.7 gigawatts, which is comparable to the power output of a nuclear power plant. Nice.



Chyurz for the knowledge